How Do Tobacco Retailers Stay Compliant in 2026?
Running a compliant tobacco retail operation in 2026 is not a single checklist you print and laminate. It is a branching set of obligations that changes based on what you sell, where you sell it, and who walks through your door. Work through each conditional below as if you are auditing your own store right now.
Start Here: Identify Your Jurisdiction’s Current Flavor Status

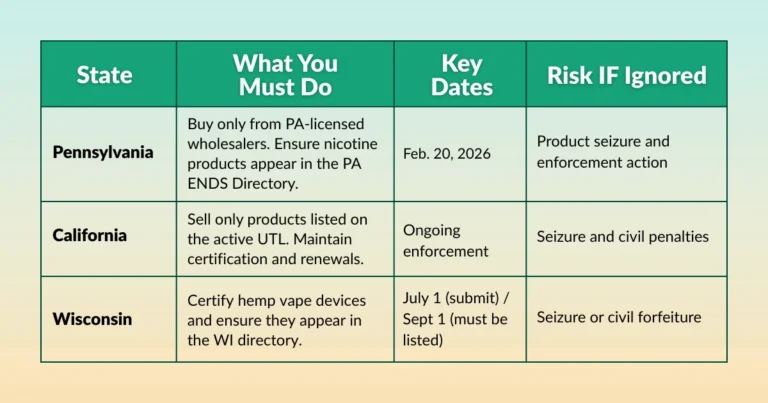
Your first compliance branch begins before you open a single package. If you sell in a state that has enacted a comprehensive flavor ban—currently including California, Massachusetts, New Jersey, and a growing list of municipalities within other states—your first step is to confirm exactly which product categories that ban covers.
Do not assume your state’s flavor ban mirrors another state’s. California’s law excludes certain hookah tobacco under licensed retailers; Massachusetts applies its ban across nearly every flavored combustible and smokeless product without carve-outs. Pull the current administrative code for your state’s flavor restriction, not a news summary of it.
If your state has no statewide flavor ban but your city or county does, you must follow the local ordinance. Local agencies—not state inspectors—will enforce it.
In that case, contact your city clerk’s office to request the current municipal tobacco ordinance in writing and confirm its effective date.This also means your product catalog cannot be globally configured. Product availability must reflect jurisdiction-specific rules at the SKU level, especially if you sell across multiple locations or ship to different regions.
If You Also Carry Nicotine Pouches

If you stock nicotine pouches—brands like Zyn, On!, or Velo—you now face a separate but overlapping compliance layer.
The FDA’s Premarket Tobacco Application (PMTA) pathway determines which nicotine pouch products are currently authorized for sale. As of early 2026, the FDA has issued Marketing Denial Orders (MDOs) for a significant number of pouch SKUs.
Your obligation is to cross-reference your current inventory against the FDA’s Tobacco Products Marketing Orders database before restocking. Selling a product under an active MDO is a federal violation, not merely a state one.
In practice, this is not just a legal check. It is an inventory and data management problem. Your product catalog, supplier feeds, and inventory systems must reflect current authorization status at the SKU level.
If the nicotine pouches you carry are authorized but flavored, and you are in a jurisdiction with a flavor ban that explicitly covers non-combustible nicotine products, you are now at the intersection of a federal authorization and a state or local prohibition. The more restrictive law governs. Remove the flavored pouch SKUs from your floor even if they carry FDA authorization.
If You Sell Vapor Products or E-Cigarettes

If your product mix includes e-cigarettes, closed-system pods, or open-system devices, your compliance tree branches further.
Every vapor product you sell must have either a PMTA authorization letter on file or a pending PMTA with a documented Refuse to Accept (RTA) or filing receipt date that keeps it in legal commerce.
If a product has no PMTA filing and is not one of the FDA’s grandfathered pre-August 8, 2016 products, it should not be on your shelf.
Many retailers rely on supplier claims or outdated documentation. Without a repeatable verification process, unauthorized products can remain active in your catalog without being flagged.
If you carry disposable e-cigarettes, specifically check whether the device exceeds 2 mL of e-liquid, as this threshold triggers scrutiny under both FDA regulations and certain state excise tax classifications. In states like Minnesota and Washington, tax obligations can depend on the nicotine concentration or container volume, not just the retail price.
This is where compliance shifts from documentation to logic. Product attributes such as volume and nicotine concentration must be accurately stored and consistently used across your tax and reporting systems.
Age Verification: Federal Floor and State Ceiling

Regardless of your state, the federal Tobacco 21 law sets the minimum purchase age at 21 for all tobacco and nicotine products.
This is your floor.
If your state’s law sets 21 as the maximum age for purchase, you comply with one standard. But several states, including Utah and certain localities, impose additional restrictions on who may purchase, when, and through what retail channel.
If you sell online or offer in-store pickup for orders placed digitally, you face the additional obligation of an FDA-compliant age verification mechanism at the point of sale, not just at checkout on a website.
The FDA’s 2024 guidance on online tobacco retail makes clear that a checkbox or self-reported birth date does not constitute sufficient age verification.
You need a verification process that can reliably confirm identity against trusted data sources and produce a defensible record of that verification.
This is not just a checkout step. It is a system requirement that must be consistently applied across channels (online, in-store pickup, and delivery)—and tied to transaction records.
If your store is within 1,000 feet of a school—a distance that varies slightly by state measurement standard—check whether your state applies additional advertising restrictions or prohibits self-service displays entirely within that radius. California and New York both do.
Inspection Readiness: What Happens When Enforcement Shows Up

Even if your product, tax, and verification systems are aligned, enforcement comes down to what you can demonstrate in the moment.
Point-of-Sale Signage
Federal law requires a “Tobacco 21” warning sign at every point of sale where tobacco products are visible or accessible.
That means:
- every register, not just your main counter
- each self-checkout terminal, if applicable
If your state requires additional signage, such as Massachusetts’ Quitline poster with specific formatting requirements, that obligation is separate and must also be met.
Signage compliance is simple—but often inconsistently applied across locations and checkout types.
If an FDA inspector or state enforcement agent visits your store, your compliance posture depends on what you can produce immediately.
You should be able to provide:
- a current vendor authorization file confirming licensed suppliers
- invoices that match your active inventory
- records of prior warning letters or inspection findings
Inspectors are not just checking your current state. They are looking for evidence of ongoing compliance and corrective action.
See How It Works in Your Environment
If you’re evaluating how to operationalize compliance across systems, Token of Trust provides a structured way to apply and verify these rules in real time—from checkout to reporting.
You can:
- validate identity and age at the point of sale
- apply rules based on product, jurisdiction, and risk level
- generate audit-ready records tied to real transactions